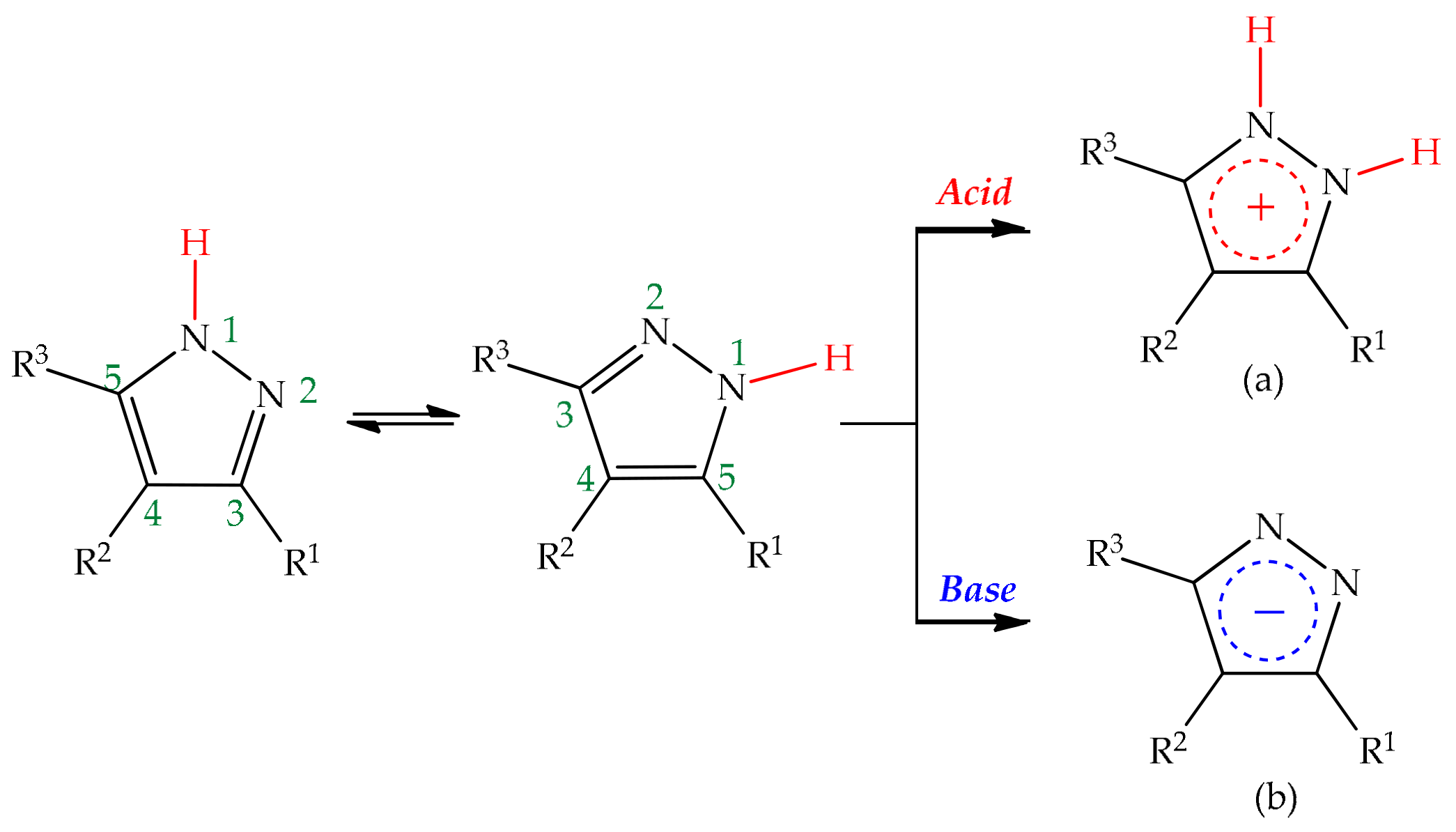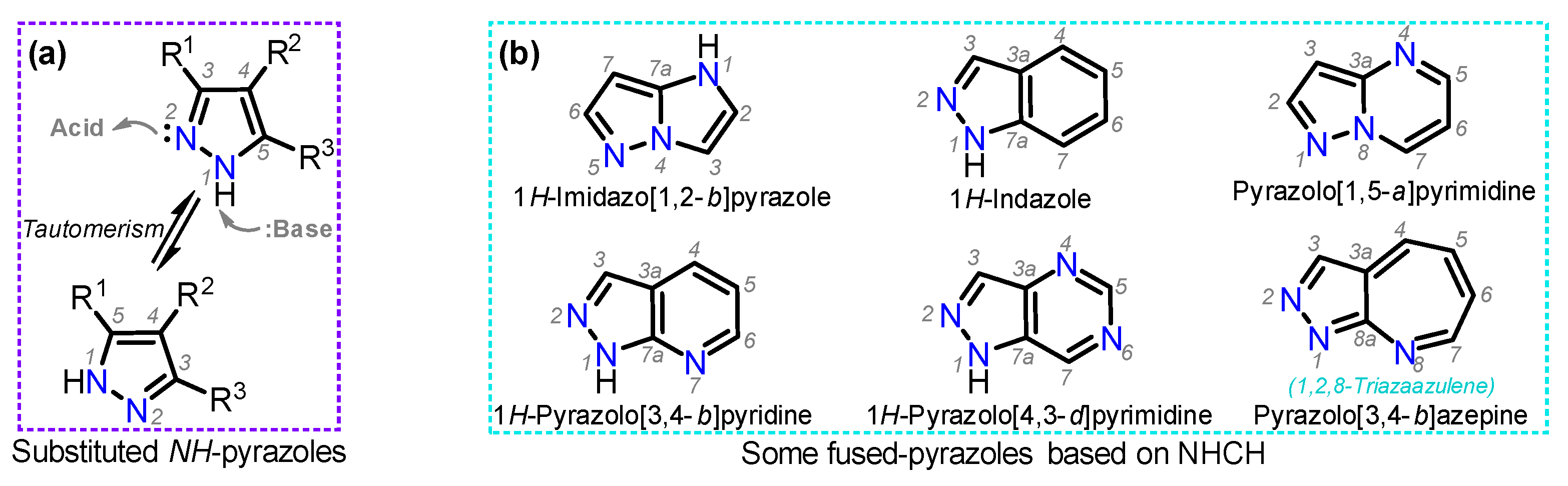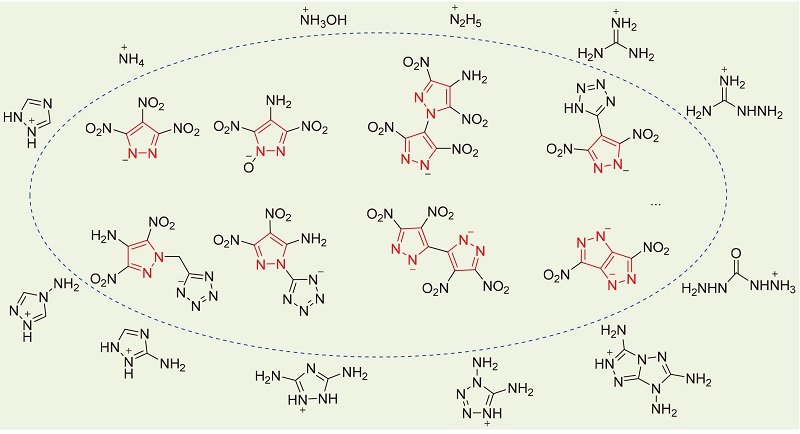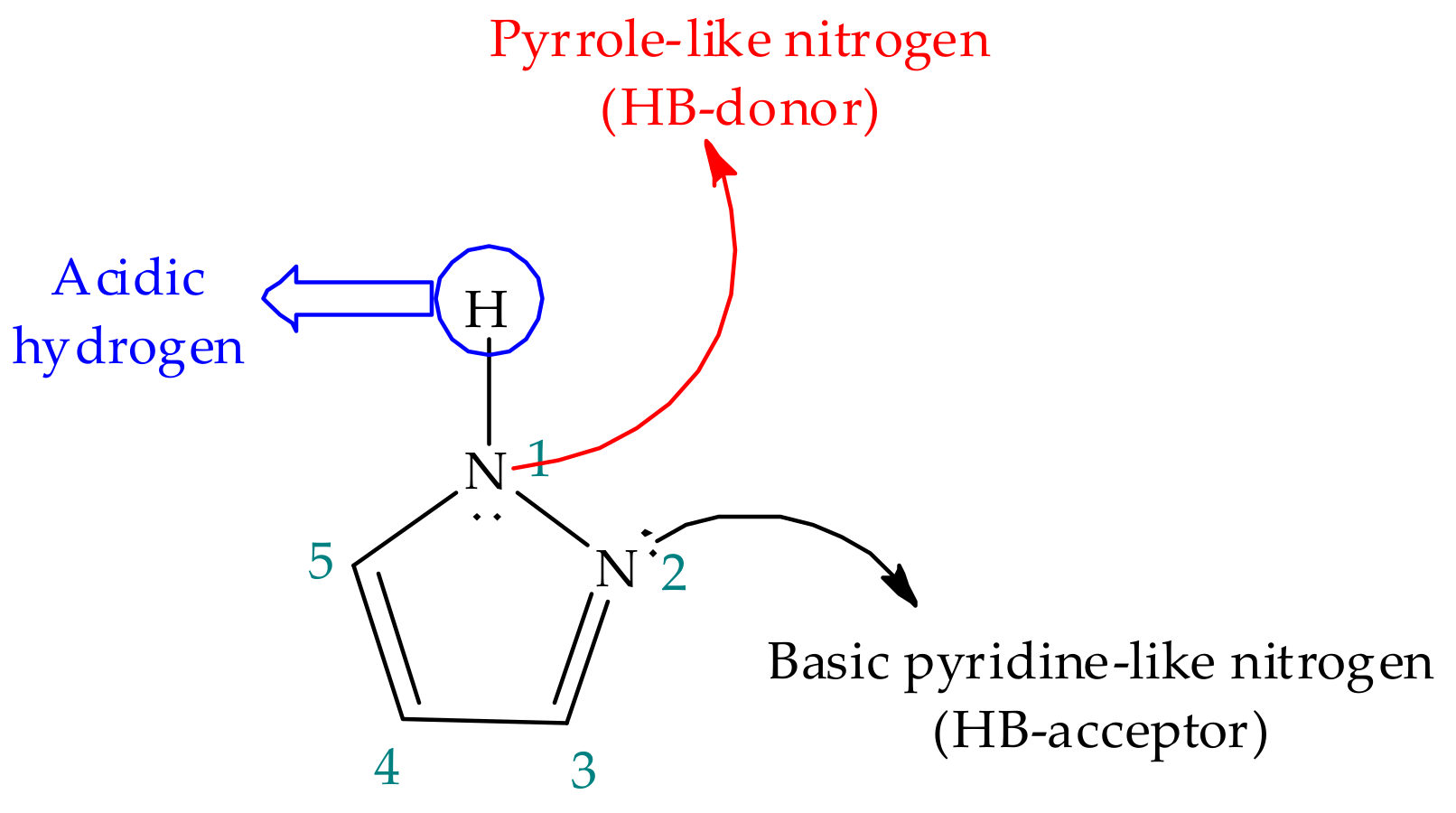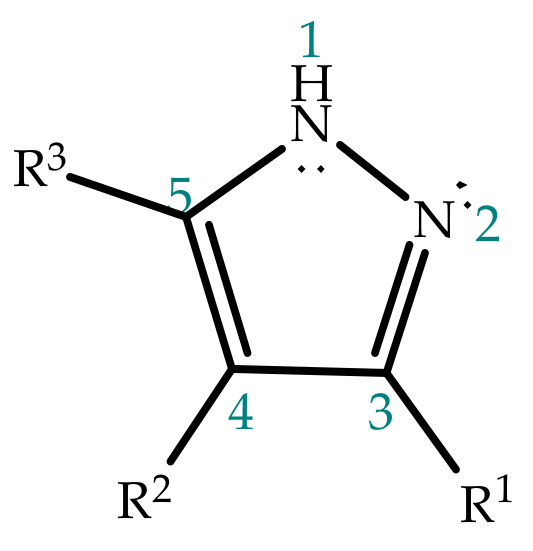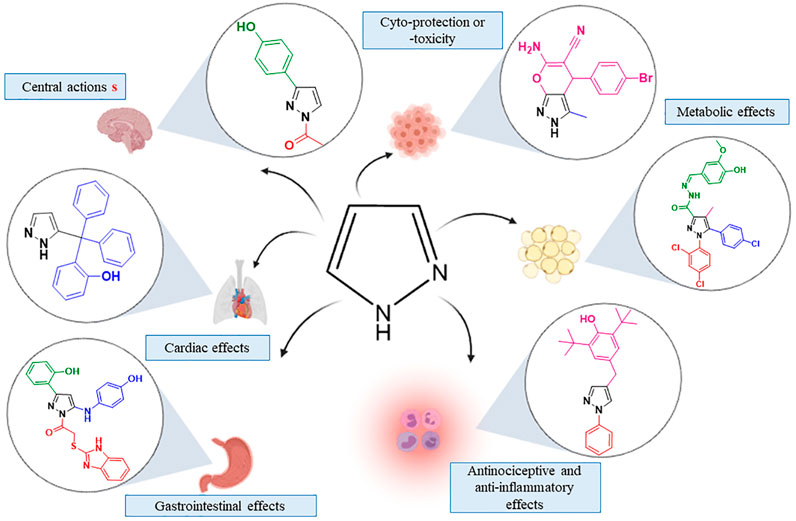
Frontiers | Heterocyclic Compounds: Pharmacology of Pyrazole Analogs From Rational Structural Considerations

Multicomponent Pyrazole Synthesis from Alkynes, Nitriles, and Titanium Imido Complexes via Oxidatively Induced N–N Bond Coupling | Journal of the American Chemical Society
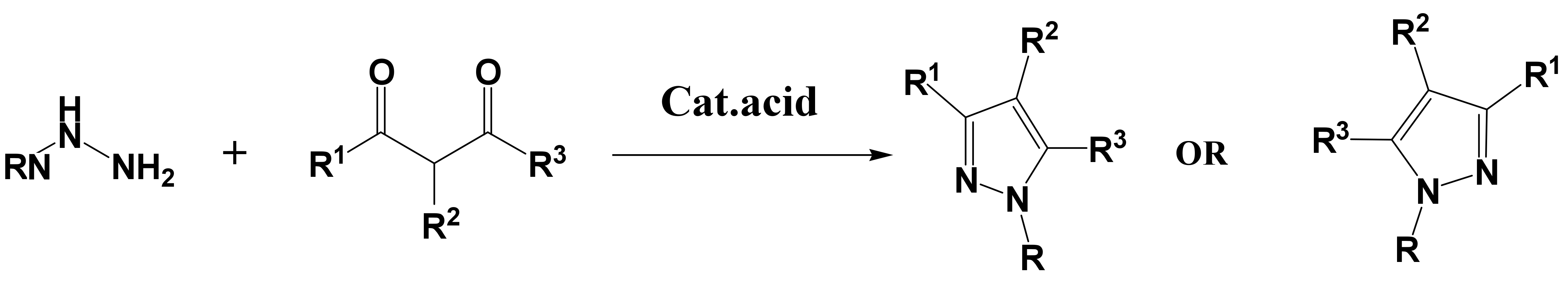
Synthesis of Nitriles – Synthesis of 4-Cyano Pyrazole, 5-Aminopyrazole Derivatives and the Deamination of 5-Aminopyrazole Derivatives | IntechOpen

Synthesis, structure, and biological activity of 4-hetaryl-2-pyrrolidones containing a pyrazole ring | SpringerLink
![BJOC - Synthesis of 2H-furo[2,3-c]pyrazole ring systems through silver(I) ion-mediated ring-closure reaction BJOC - Synthesis of 2H-furo[2,3-c]pyrazole ring systems through silver(I) ion-mediated ring-closure reaction](https://www.beilstein-journals.org/bjoc/content/figures/1860-5397-15-62-share.png)
BJOC - Synthesis of 2H-furo[2,3-c]pyrazole ring systems through silver(I) ion-mediated ring-closure reaction

Structural Insights into Pyrazoles as Agents against Anti‐inflammatory and Related Disorders - Priya - 2022 - ChemistrySelect - Wiley Online Library
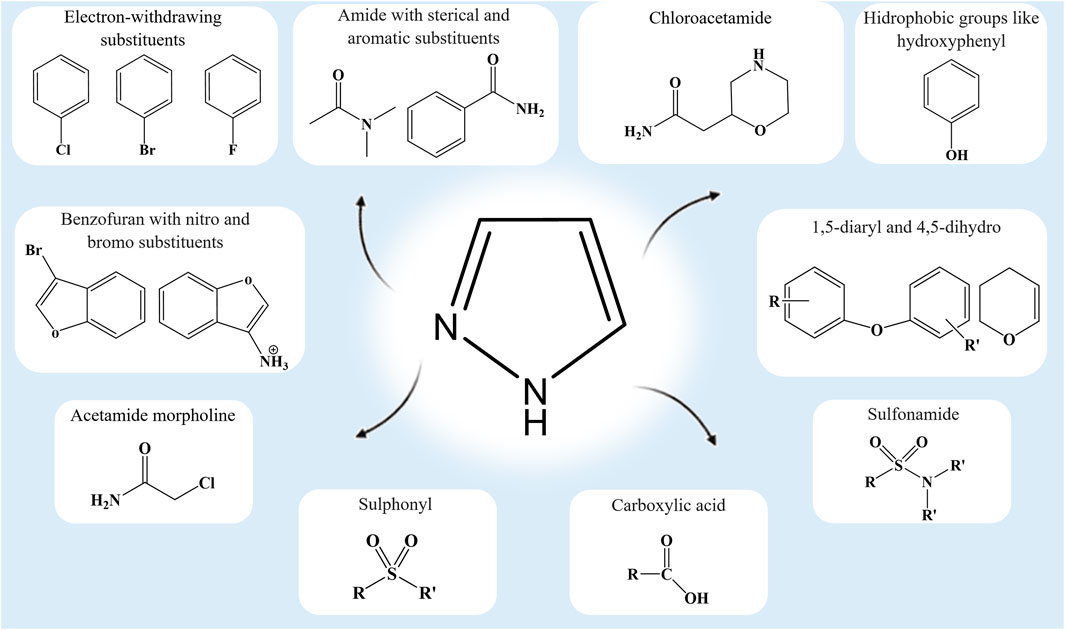
Frontiers | Heterocyclic Compounds: Pharmacology of Pyrazole Analogs From Rational Structural Considerations

Unified Access to Pyrimidines and Quinazolines Enabled by N–N Cleaving Carbon Atom Insertion | Journal of the American Chemical Society
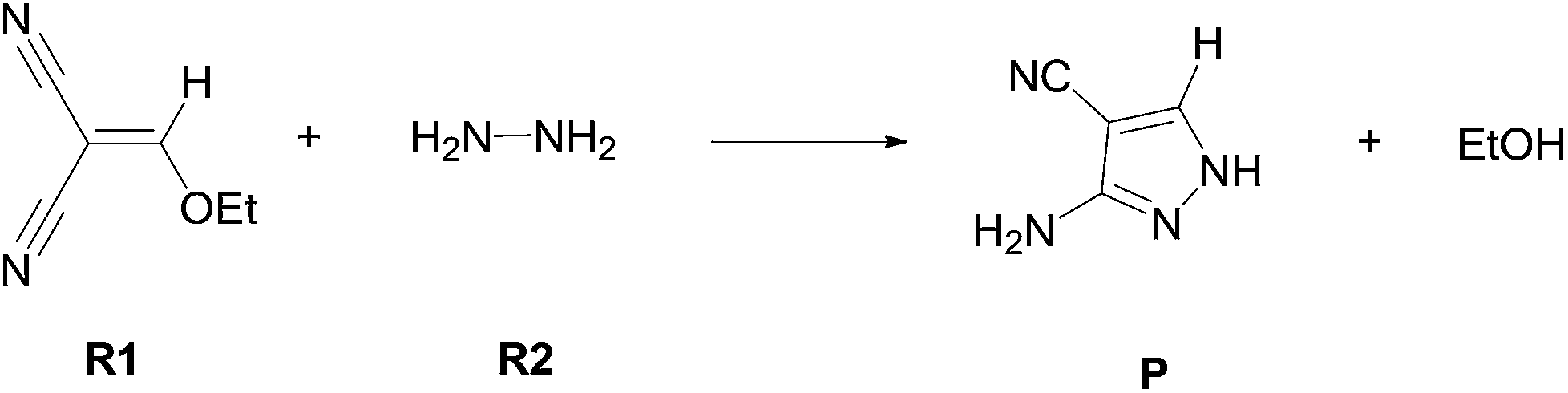
Theoretical investigation of the chemoselectivity and synchronously pyrazole ring formation mechanism from ethoxymethylenemalononitrile and hydrazine ... - RSC Advances (RSC Publishing) DOI:10.1039/C4RA06316C

Elucidating the Mechanism of Cytochrome P450–Mediated Pyrimidine Ring Conversion to Pyrazole Metabolites with the BACE1 Inhibitor GNE-892 in Rats | Drug Metabolism & Disposition